Facebook Comment Change Revives Pharma Call for FDA Guidelines
For pharma brands, the FDA is the elephant in Facebook comment room.
For pharma brands, the FDA is the elephant in Facebook comment room.
On Monday, Facebook began requiring pharmaceutical brand pages to enable comments, as expected. Yet, although pharma marketers are fearful of regulatory reaction to a potential flood of comments, some don’t see Facebook as the big villain in the story. It’s the Food and Drug Administration that’s raised the most ire.
“Yesterday’s announcement about Facebook’s new policy only served to underscore the importance of the FDA’s long-time forthcoming guidance,” wrote Kate Conners of The Pharmaceutical Manufacturers Association on the trade group’s blog yesterday.
“What’s more, the FDA has not clarified whether companies should be held responsible for the comments on third parties posted on company-controlled Web sites; the government promised a relevant guidance on these issues in 2009.”
 Pharma companies have been begging the FDA to give guidance on use of social media communications by pharmaceutical brands since before a hearing on the issue was held in November 2009. Though the FDA’s Division of Drug Marketing, Advertising, and Communications – the body which reviews ads for prescription drugs – has said it plans to release guidelines on at least one related topic this year, the first quarter 2011 deadline it last gave has come and gone.
Pharma companies have been begging the FDA to give guidance on use of social media communications by pharmaceutical brands since before a hearing on the issue was held in November 2009. Though the FDA’s Division of Drug Marketing, Advertising, and Communications – the body which reviews ads for prescription drugs – has said it plans to release guidelines on at least one related topic this year, the first quarter 2011 deadline it last gave has come and gone.
“One thing we’re expecting [the Facebook change] to do is to put a little bit more pressure on the FDA to come up with something,” said Monique Levy, VP of research at health and pharma industry research firm Manhattan Research. “It’s giving the whole issue renewed attention.”
Starting Monday August 15, Facebook now requires pharma brands to include comments on their pages. Since Facebook announced the upcoming switch in May, pharma marketers have been scrambling to come up with solutions that satisfy their colleagues in legal. Most brands have tiptoed into social media environments like Facebook and Twitter, recognizing the potential marketing value, but remain extremely cautious.
The Facebook changeover has disrupted the no-comment cocoons pharma brands have snuggled in until now, and firms are taking a variety of safe approaches to navigating the newly conversational landscape of their brand pages.
“We want to make sure that we’re monitoring on a regular basis throughout the day to ensure that anything that’s up there we’re able to respond to appropriately,” said Stacy Burch, director of corporate reputation and digital communications at Sanofi in the U.S. in an August 15 interview with Washington, D.C. lawyer Mark Senak featured in a podcast on his EyeOnFDA blog. The company has kept its Sanofi-aventis U.S. Diabetes page intact, which now includes comments. “The absence of any official FDA regulations” is the main issue for pharmaceutical companies, said Burch.
In his interview, Senak referred to August 15 as “D-Day” and “a notorious day for those of us in digital communications.”
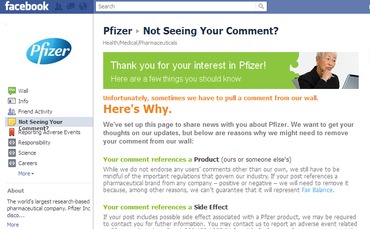
“Now that you can respond to our posts, please know that we may sometimes need to remove a comment. Click on the ‘Not Seeing Your Comment?’ link on the left to learn why. Thank you for visiting us!” wrote Pfizer on its corporate Facebook page on Monday. The company also added a new “not seeing your comment?” tab on the left-side page navigation.
Pharma marketers are required to report adverse effects of their drugs, so if someone posts a comment about an adverse effect on a Facebook page, the company is responsible to report that to the FDA. Also, when they become aware of online conversations including incorrect or off-label information about their drugs and products, they need to notify the agency.
In a lengthy explanation on Facebook, Pfizer detailed “why we might need to remove your comment from our wall.” The company listed several reasons including, “Your comment references a Side Effect,” and “Your comment contains Medical Advice.” Another reason for takedown: “If your post references a pharmaceutical brand from any company – positive or negative – we will need to remove it because, among other reasons, we can’t guarantee that it will represent Fair Balance.”
Other pages remain alive with comments, including Sounds of Pertussis, an education project from Sanofi Pasteur Inc. and March of Dimes; The Diabetes Dish, a Merck page; and Pfizer Hemophilia’s Hemophilia Community.
In some cases, comments could become a blessing in disguise, fostering the relationships brands supposedly developed Facebook pages for in the first place. “You are doing a fantastic thing by spreading awareness,” wrote a commenter on the Sounds of Pertussis page. “My almost 2 month old son is now a pertussis survivor…. I have been doing everything I can to tell everyone I know our story. I’ve been linking people to your site, this page & the digital quilt.”
 Meanwhile, the Facebook decision appears to have been a death sentence for some pages. Allergan’s Juvederm, Latisse, and Join the Millions – a Botox Cosmetic page – all feature this line alone on their walls: “There are no more posts to show.”
Meanwhile, the Facebook decision appears to have been a death sentence for some pages. Allergan’s Juvederm, Latisse, and Join the Millions – a Botox Cosmetic page – all feature this line alone on their walls: “There are no more posts to show.”
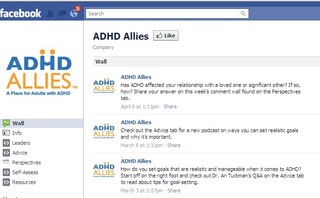
Some pages have been wiped from existence. For instance, as reported by Jonathan Richman on his Dose of Digital site, where he’s been tracking the impact of the Facebook changeover, ADHD Allies, a page from McNeil Pediatrics, has been removed.
Update: After this story was published, the wall of Merck’s “The Diabetes Dish” page on Facebook made no posts available.
Leave a Reply
You must be logged in to post a comment.